Equation for epidemic spread with quarantine measures: application to COVID-19
Beitrag zur Diskussion in der Leibniz-Sozietät
The appearance of a new virus in China at the end of 2019 has been recognized by the World Health Organization (WHO) as a pandemic in March 11, 2020. This virus named SARS-CoV-2 (causing COVID-19 disease) to the moment infected 50 millions of people and more than 1 million of deaths over the world. High mortality required introduction of essential quarantine measures to confront pandemic and to reduce victims. At the same time, the strong quarantine measures lead to the serious damage to the global economy.
Soon for a year now, the world has been living in a completely new regime, trying to minimize the victims of the epidemic, economic damage and manifestations in a number of countries of overt or covert protests associated with different attitudes towards the problem in different layers of society. This situation can be called the New Covid Reality (NCR), so deeply and everywhere it changed people’s life.
At the same time, the NCR will certainly accelerate the development of new methods in medicine, biology, education, business and will lead, hopefully, to a new, more realistic, adequate and humanistic understanding and solution of political problems.
A huge efforts have been made to accelerate the development of effective COVID-19 vaccines and to prepare the capacity for their production. In Russia, for the first time (albeit with a change in the accepted registration procedure after the third stage of testing), the Sputnik V vaccine has been registered and is undergoing the last stage of testing; many other vaccines have been created and tested. The American pharmaceutical company Pfizer and German biotechnology company BioNTech just reported that their vaccine is more than 90% effective at preventing COVID-19. In England, there is a group of volunteers who agree to artificially infect COVID-19 after vaccination and the formation of a sufficiently high level of antibodies in order to test the effectiveness of the AZD 1222 vaccine. Apparently, permission to conduct such unique trials aimed at quickly testing the effectiveness of the vaccine will be given.
In the clinical treatment of patients, the effects of existing and new medications are studied.
The most effective anti-epidemic means and measures to protect people, including new methods based on Internet technologies and mobile communications, are being investigated and identified. Many scientific and scientific-technical conferences, seminars and sessions are held online (zoom or meet). The same applies to the educational process in many universities and schools.
New methods for studying epidemic processes that are more adequate to the specifics of COVID-19 are also being developed.
Analytical and computer models are being created that take into account the specific features of the transmission of the SARS-CoV-2 virus and the spread of COVID-19.
The most of existing models for the spread of infection simulate the spontaneous development of an epidemic and describe all its stages. There are two kinds of such models: susceptible-infected-susceptible (SIS) models and susceptible-infected-recovered (SIR) models. The first go back to the pioneering work of Kermack and McKendrick [1] and uses the assumption that the recovered people can immediately get infection again. On the contrary, the SIR model is built on the assumption that the recovered people save strong immunity during epidemic (see, e.g. [2]). Many variants of the SIS [3] and SIR, e.g. [4,5], models are used in the mathematical epidemiology.
The susceptible-infected-recovered (SIR) models assume the overall immunity of recovered people and opportunity of immediate recovery. However, the last assumption is quite questionable for diseases like COVID-19.
In the recently proposed discrete model of epidemic [6,7] we considered some of specific features of COVID-19. Considering a closed population, the model includes a few independent parameters. One of them is the average duration of disease d, and the second, the parameter of the infection transmission rate p, similar to the reproduction number R0 in the SIR models. We divide the parameter p on the product of two n* k, where n is the average quantity of dangerous contacts per day for one infected person and k is the average susceptibility to virus of a healthy person. The initial condition, namely the quantity of the infected people at the beginning or for some developed stages of the epidemic process in the concrete closed region, is also essential.
The national and local authorities around the world take action to slow down the epidemic. We can define these quarantine measures as an epidemic under partial control. It is very important to slow down the epidemic rate to give physicians the ability to provide patients with the necessary amount of medical care.
We believe that it is important to take into account such features of the disease caused by COVID-19, as the frequent recurrence of asymptomatic course of the disease, a long disease duration, high infectivity and high mortality. The model we presented in [6,7] makes it possible to take into account all features just mentioned. We consider the course of epidemic in the closed population. Based on this, the authorities can outline a strategy for quarantine measures.
If there is no special quarantine measures the epidemic course is free. One of the main futures of the free epidemic course is the existence of the collective immunity, namely the maximum percentage of cases in the population. The model for COVID-19 [6] predicts the collective immunity for the free epidemic course as the function of the average parameters p and d (see the examples on Figure 1).
The main result of this work is the new equation for an epidemic process description in a closed population, which takes into account the quarantine measures. Following to the available statistical data [8] we formulated the epidemic model in discrete time (let say, days l, day by day) for two functions NT(l) equal to total cases in a close region and NI equal to the current number of virus carriers, which are infectious for the healthy people. These equations are [6]:
NT (l)=NT(l-1)+NI(l-1)*{1+p(l)*[1-NT(l-1)/N] } ,
NI (l)=NT(l)-NT(l-d+1).
From these two equation we find the closed equation for NT (l):
NT (l)=NT(l-1)+[ NT(l)-NT(l-d+1)]*{1+p(l)*[1-NT(l-1)/N]}.
This equation can be characterized as the delayed iterative equation. Here N is the full number of people in the considering population and p(l) is the „function of external influence“, which takes into account introduction and influence of the quarantine measures. For the first (free) stage of epidemic (the free stage is various for different countries, usually of order 30 days) the value p(l)=const=p can be found by the reciprocal count by use the official statistical data [8]. The nonlinearity in equation for NT(l) is related with the impossibility to transfer infection from the virus carriers (infectious) to themselves and to recovered (and dead).
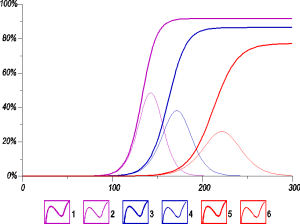
Fig. 1 The total number of infections (thick curves) and the number of actually infected (thin curves) computed on the basis [6] during the free-running epidemic in a closed populations a percentage of the population size. Here p = 0.14 (violet curves 1, 2), p = 0.12 (blue curves 3, 4), p=0.1 (red curves 5, 6); d = 20.
For the next period p(l) describes the gradual reduction of dangerous contacts due to special quarantine measures (masks, gloves, sanitizers, restrictions in various business, travelling, transferring to online education and so on) of the epidemic COVID-19.
The function p(l) is unknown a priory and can be found empirically to describe the statistical data. In future the influence of various quarantine measures can be determined sufficiently exact to calculate p(l).
The closed nonlinear delayed equation for NT (l) can be easily solved numerically.
The results are shown on Figures 2, 3 for Germany.
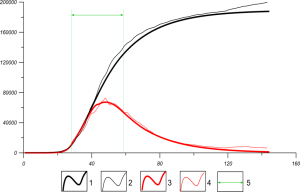
Fig. 2 Epidemic course of COVID-19 for Germany; x-axis – l, or days after the first infection, y-axis – the cases of viral infection, data for first 140 days; N = 8*107, d=16; 1 (black thick line) – total cases computed on the basis of equation for NT (l) [6]; 2 (black thin line) – total cases observed [8]; 3 (red solid line) – active cases computed on the basis of equation for NI (l) [6]; 4 (red thin line) – active cases observed [8]; 5 – the green arrow marks „the hyperbolic approximation“ for the function of external influence p(l)=0.0348+1.28163* 105 / l4 between l0 = 28 and l=58; p(l)=0.403 for days l0 <l=28 and p(l>58)=p(58)=0.046.
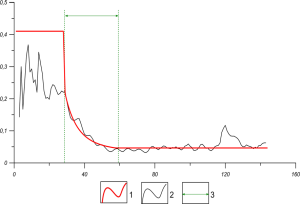
Fig. 3 Germany, p(l) with the hyperbolic sector between the days l=28 and l=58, p(l)=0.0348+1.28163*105/ l4 . Here 1 (red line) – the approximation applied to p(l) [6], 2 (black line) – the observed p(l) [8], 3- green arrow – days for the used „hyperbolic approximation“.
In [6] the similar calculations based on the model are performed also for Austria with population 8*106 and for Israel with approximately the same population 8*106. For these countries a good agreement between the statistical data and calculations based on the theoretical model has been found for the „hyperbolic approximation“ of p(l)= A+B/ l4 with different constants A and B. In general, the dependence p(l) can be different, depending on many factors, including the subordination of people to the official rules. The graduate reduce of the „function of an external influence“ obeys also to graduate inclusion of people to the quarantine measures. The transferring from discrete time model to the delayed differential equation [7], which is the generalized Lotka-Volterra equation [9,10], shows the essential difference in comparison with the SIR model due to the delay obliged by the long duration of COVID-19. The earlier the restricted quarantine measures are introduced and the stricter they are implemented and monitored, the easier it is to achieve the collective immunity under quarantine measures at a relatively low level 0.2%-2% of population and even less [7]. This is in line with both the results of the developed model and the practical results achieved, for example, in China, where the pandemic began, and in S. Korea, where it was practically suppressed due to first extremely strict, and now very moderate quarantine measures.
Necessary to stress, that for description of so called „second wave“, which is in fact the new splash of epidemic due to weakening of quarantine measures, an another form of the „function of external influence“ p(l) should be introduced. However, the presented theory can describe all the process, including periodical change of the balance between quarantine measures and reduction of the quarantine restrictions.
References:
[1] Kermack, W. O., and McKendrick, A. G., A contribution to the mathematical theory of epidemics. Proc. Royal Soc. A 115, 700–721 (1927)
[2] Brauer, F.; Castillo-Chavez, C., Mathematical Models in Population Biology and Epidemiology. Springer-Verlag, 2000
[3] Ball, F., Stochastic and deterministic models for SIS epidemics among a population partitioned into households, Math. Biosci., 156, 41 (1999)
[4] Sander L.M., C. P. Warren C.M., Sokolov I.M., Epidemics, disorder, and percolation Physica a: Statistical Mechanics and Its Applications, 325, 1 (2003)
[5] Murray J.D., Mathematical Biology, Springer Verlag, New York, 1993.
[6] Trigger S.A., Czerniawski E.B., Equation for epidemic spread with the quarantine measures: application to COVID-19, Physica Scripta 95 105001 (2020).
[7] Trigger S.A., Czerniawski E.B., Ignatov A.M., The model for epidemic transmission with quarantine measures: application to COVID-19, Research Gate, July 2020; paper in print.
[8] Worldometer counter, https://www.worldometers.info/coronavirus.
[9] Lotka A. J., Elements of Physical Biology. Williams and Wilkins (1925)
[10]Volterra V., Variazioni e fluttuazioni del numero d’individui in specie animali conviventi, Mem. Acad. Lincei Roma. 2: 31–113 (1926).
Sergey Trigger, MLS
EMail des Autors: satron@mail.ru
Siehe dazu auch die Jahrestagung der Leibniz-Sozietät 2019
zum Thema
„Virusinfektionen – alte und neue Erreger sowie Wege der Impfprophylaxe“